Similar Posts
Union of sets
Union of sets Let A and B be two sets. The union of A and B is the set of all those elements which belong either to A or to B or to both A and B. We denote A union B by notation “A ∪ B” Thus A∪B = { x : x ∈…
Subsets of the Set R of Real Numbers
Subsets of the set R of real numbers Following sets are important subsets of the set R of all real numbers: (i) The set of all natural numbers N = { 1, 2, 3, 4, 5, 6,…. } (u) The set of all integers Z = { … – 3, – 2, -1,…
About Quasi-static process
Quasi-static process A process during which the system remains nearly close to an equilibrium state is called Quasistatic process. In other words departure of the state of a system from Thermodynamic Equilibrium state will be Infinitesimally small. Quasi-static process Consider a system as shown in figure the system initially is in equilibrium state as weights…
Intervals as Subsets of R
Intervals as subsets of R Closed intervals Let a and b be two given real numbers such that a < b. Then the set of all real numbers x such that a ≤ x ≤ b is called a closed interval and is denoted by [a, b] . Thus, [a, b] = {…
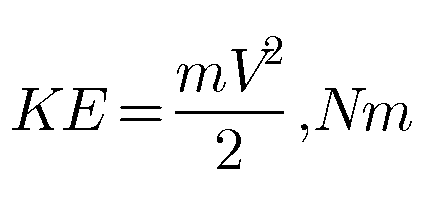
Energy in a State | Point and Path Functions in Thermodynamics
Energy in a state Stored energy is the energy possessed by a system. Stored energy in the system is called energy in a state. It is associated with a state. It change depends only on the end states of the process and not on the path of…
Complement of a Set
Complement of a set Let U be the universal set and let A be a set such that A ⊂ U. Then the complement of A with respect to U is denoted by A’ or Ac or U-A and is defined the set of all those elements of U which are not in A. Thus…

Nice
Nice
Nice