Properties of Ideal gases ∣ Charactaristic of gas equation ∣ Specific Heat
Ideal gases
Boyle and Charles investigated the behaviour of gases and formulated the laws.
Quasi-static process A process during which the system remains nearly close to an equilibrium state is called Quasistatic process. In other words departure of the state of a system from Thermodynamic Equilibrium state will be Infinitesimally small. Quasi-static process Consider a system as shown in figure the system initially is in equilibrium state as weights…
Semi-Open or Semi-Closed interval If a and b are two real numbers such that a < b, then the sets (a, b] = { x : x ∈ R, a < x ≤ b} and [a, b)={ x 😡 ∈ R, a ≤ x < b are known as semi-open or semi-closed intervals . (a,…
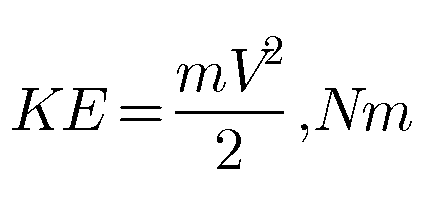
Energy in a state Stored energy is the energy possessed by a system. Stored energy in the system is called energy in a state. It is associated with a state. It change depends only on the end states of the process and not on the path of…
Macroscopic and Microscopic Approaches The behaviour of matter can be studied by two approaches 1. Macroscopic approach, 2. Microscopic approach 1. Macroscopic approach – In the macroscopic approach a certain quantity of matter is considered for study without knowing the behaviour of individual…
Relation Let A and B be two sets. Then a relation from set A to B is a subset of A × B. Thus, R is a relation from A to B⇔R ⊆ A × B. If R is a relation from a non-void set A to non-void set B and my if (a, b)…
Union of sets Let A and B be two sets. The union of A and B is the set of all those elements which belong either to A or to B or to both A and B. We denote A union B by notation “A ∪ B” Thus A∪B = { x : x ∈…
Hello
How can i help you